We are open to partnering, contact our team at
Phase 2 Ready orally administered p38 MAP Kinase inhibitor – a preventative therapy targeting an addressable market of more than $10 billion.
POLB 001 aims to target the Cytokine Release Syndrome (CRS) associated with many cancer immunotherapy treatments which impacts >70% of patients undergoing CAR T or Bispecific Antibody therapy. Severe cases of CRS are life-threatening and may require intensive supportive care. Mild to moderate CRS can result in extended hospitalisation and high consumption of healthcare resources.
Rapid advancements in cancer immunotherapy are driving demand for effective CRS treatments. POLB 001 has the potential to positively impacting patients and healthcare systems worldwide by enabling broader, safer delivery of these therapies to the cancer patients who need them.
Symptoms of CRS

Compelling Data
- Phase 2 ready oral small molecule, excellent bioavailability
- Strong pre-clinical data package
- Proven safety profile & well tolerated in Phase I clinical trial
- Efficacy demonstrated in Phase 1b human challenge trial supports partnering
- Efficacy demonstrated in reducing cancer immunotherapy-induced CRS in an in vivo model
Strong Patent Portfolio
- Oncology patent applications filed 2023, potential for protection out to 2043 – recent data enhances & facilitates expansion of patent applications
- Granted patents for severe influenza to 2038
Major Market Opportunity
- Significant market opportunity in cancer immunotherapies (e.g. CAR T, bispecific antibody therapies) which are expected to reach c. $120bn by 20301-3
- CRS is a barrier to broader uptake of cancer immunotherapies, ttolarge pharma seeking a product differentiator
- POLB 001 potential market >US$10 billion
- CRS induced by cancer immunotherapies has the potential to be a rare / orphan indication

1. Grand View Research. CAR T-Cell Therapy Market Analysis 2023-2030. 2. Grand View Research. Bispecific Antibodies Market Size,Share & Trends Analysis Report. 3. Datamonitor Healthcare. Forecast: Diffuse Large B-Cell Lymphoma and Multiple Myeloma, 2023.h
Clinical trial enabling activities to advance POLB 001 in oncology are progressing.
Watch the POLB 001 Oncology Programme Update to learn more
Data Presented at the 65th American Society of Hematology (ASH) Meeting, learn more

Abstract #189062
POLB 001, an oral broad spectrum anti-inflammatory with the potential to prevent Cytokine Release Syndrome (CRS)
Existing Data on POLB 001: Human LPS Challenge Trial
Evidence for benefit of POLB 001 in the therapy of LPS-induced inflammation
Supplementing the applicability of POLB 001 to severe influenza on the one hand and immunotherapy-induced CRS on the other, POLB 001 has been investigated in a human lipopolysaccharide (LPS) challenge trial.
Randomised, double-blind, placebo-controlled, multiple dose, inflammatory challenge trial in healthy volunteers
Trial design

Endpoints
Intravenous LPS challenge
- Bloods (cytokines, vascular markers, CRP)
- Ex-vivo LPS response
- Safety & tolerability (inc. vital signs, AE’s, ECG, Haematology)
- Local inflammatory responses were also measured
Results Demonstrate POLB 001 can Potently Inhibit Inflammation
POLB 001 was widely distributed, reduced the inflammatory response and inhibited p38 MAPK activation and signaling following LPS challenge.
- Excellent safety profile across two clinical studies
- Potent target inhibition confirmed
- Major reduction of key inflammatory markers
- Clear dose response relationship observed
Potent and Selective Inhibition of p38 MAPK Signaling
Effective target engagement demonstrated

Levels of phosphorylated p38 MAPK in circulating monocytes
- POLB 001 was widely distributed
- POLB 001 inhibited p38 MAPK activation, direct measurement of activation
- POLB 001 inhibited in vivo and ex vivo responses to LPS-induced TNF-α, indirect measurement p38 activity
Blood samples were taken before and after administration of intravenous LPS. Peripheral blood samples were analyzed by flow cytometry. Monocytes were gated by FSC, SSC and CD14+. Data is presented as mean MFI values of phospho-p38 +/- SEM
Reduced Key Inflammatory Cytokines Following LPS Challenge
Dose dependent reductions of inflammation
TNF-α

TNF-α reduction of 73.5% and 56.2% seen for 70 mg and 150 mg doses respectively (p = 0.0003†)
IL-6

IL-6 reduction of 57.4% and 63.5% seen for 70 mg and 150 mg doses respectively (p = 0.0002†)
IL-8

IL-8 reduction of 80.7% and 76.7% seen for 70 mg and 150 mg doses respectively (p < 0.0001†)

TNF-α, IL-6 and IL-8 levels decreased between 56-81% in subjects treated with 70 mg or 150 mg POLB 001 twice daily
†The exploratory analysis suggested statistically significant improvement in treatment (p<0.05) for the endpoints examined.
Reduced Key Indicators of LPS-Induced Systemic Inflammation
The reduction of systemic cytokines aligns with improvement in clinically meaningful endpoints
Mean Body Temperature

No significant effect on body temperature with a trend towards reduction compared to placebo.
Heart Rate Rise (bpm)

Suppressed increase in heart rate following IV LPS administration
C-Reactive Protein (CRP)

CRP level reductionof 33.1% and 33.3% seen for 70mg and 150mg doses respectively

POLB 001 Effectively Reduced Inflammation in Tissue
POLB 001 150 mg significantly reduced IL-1β† and TNF-α† responses in blister exudate compared to placebo
TNF-α in blister exudate

IL-1β in blister exudate
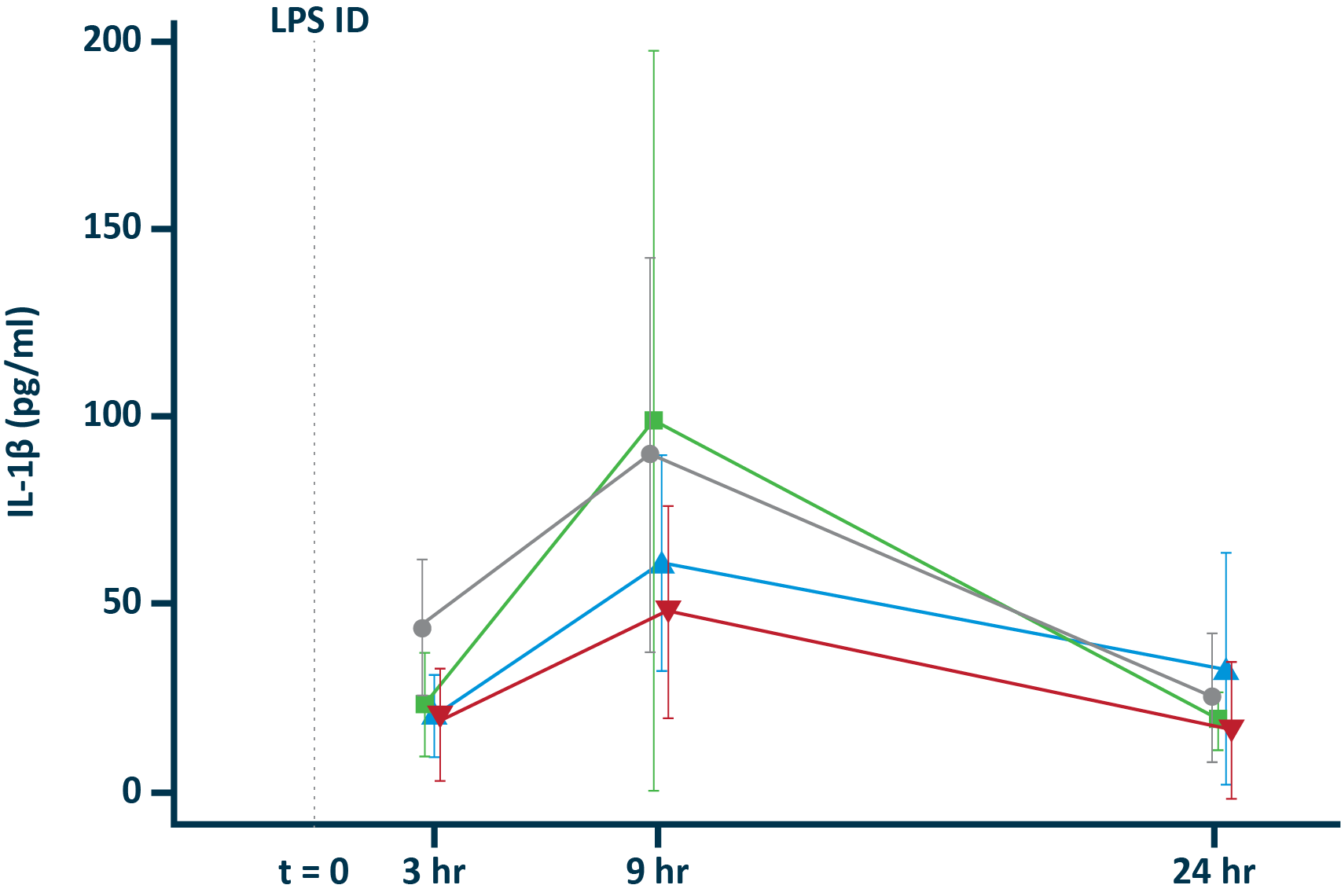
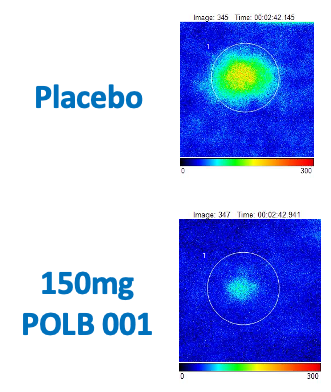

†The exploratory analysis suggested statistically significant improvement in treatment (p<0.05) for the endpoints examined.
