We are open to partnering, contact our team at



CRS: Cytokine releases Syndrome. MAPK: Mitogen Activated Protein Kinase. TNF: Tumour necrosis factor, IL-6: Interleukin-6, TOPICAL – Trial of Prevention of ImmunoCytokine Adverse events in Myeloma
Effective preventative therapy represents a market opportunity exceeding US$10B




Navigate POLB 001:
Significant Market Opportunity in a Rapidly Growing Field
CRS is a major issue and rate limiting in delivering cancer immunotherapies
- No approved therapy for CRS prevention & few approved for CRS management
- The need for effective CRS management is being driven by rapid growth of CRS-inducing immunotherapies
- Bispecific antibody and CAR T therapy market expected to grow exponentially
Bispecific Antibodies & CAR T Therapies4,5,6

POLB 001 – potential first approved preventative therapy for cancer immunotherapy-induced CRS
Significant opportunity exists for POLB 001 as CRS preventative for bispecific antibody & CAR T treatment3
•1st, 2nd and 3rd line+ MM and DLBCL patients in the US and EU5, may become eligible to receive CAR T and bispecific antibody therapy11
•An effective preventative therapy for CRS could enable outpatient administration and broader uptake of immunotherapies12
•Potential across additional haematological malignancies, solid tumours and new areas like severe influenza
•The cost to treat a patient with grade 3 CRS can be >$70k12

Potential to Greatly Enhance Uptake of bispecific antibody and CAR T Therapies
Effective prevention of CRS by POLB 001 may enable broader access to cancer immunotherapies

*CRS prevention may contribute to bottleneck removal. Other issues, such as manufacturing, supply and other adverse events, may also present barriers to wider uptake.
Independent Payer Research Covering c.75M Lives Across Commercial Insurance, Medicare & Medicaid
Multi-billion $
POLB 001 peak
sales potential
“This research validates both the scale of that unmet need and the appetite among payers for an effective preventative approach. We observed a willingness to pay at commercially meaningful price points, driven by the potential to reduce hospitalisation costs, potentially allowing for care to be decentralised and ultimately delivering better outcomes for some of the sickest patients. This positions POLB 001 as a compelling CRS solution with significant market potential.“
Chris Grimes Crompton, Partner, Value Strategy, Acumetis Global
Key Opinion Leaders Supportive of POLB 001’s Significant Potential
Dr Martin Kaiser, Consultant Haemato-Oncologist The Institute of Cancer Research & Royal Marsden Hospital discusses CRS and its impact on cancer immunotherapy administration
August 2025
“Bispecific antibodies will only be delivered in specialist cancer centres until there is a way to make treatment safe enough to extend them to a much wider patient population.”
Prof Gareth Morgan, myeloma specialist, US
“If there was a therapy that was orally delivered, a whole lot of infrastructure requirement falls away.”
Prof Martin Kaiser, myeloma specialist, UK
POLB 001 Scientific & Supporting Data
Potent and Selective Inhibition of p38 MAPK Signaling
Effective target engagement demonstrated in LPS human challenge trial


Levels of phosphorylated p38 MAPK in circulating monocytes
- POLB 001 was widely distributed
- POLB 001 inhibited p38 MAPK activation, direct measurement of activation
- POLB 001 inhibited in vivo and ex vivo responses to LPS-induced TNF-α, indirect measurement p38 activity
Blood samples were taken before and after administration of intravenous LPS. Peripheral blood samples were analysed by flow cytometry. Monocytes were gated by FSC, SSC and CD14+. Data is presented as mean MFI values of phospho-p38 +/- SEM
Reduced Key Inflammatory Cytokines Following LPS Challenge
Dose dependent reductions, without ablation of immune system, leaving key immune functions intact

TNF-α

TNF-α reduction of 73.5% and 56.2% seen for 70 mg and 150 mg doses respectively (p = 0.0003†)
IL-6

IL-6 reduction of 57.4% and 63.5% seen for 70 mg and 150 mg doses respectively (p = 0.0002†)
IL-8

IL-8 reduction of 80.7% and 76.7% seen for 70 mg and 150 mg doses respectively (p < 0.0001†)

TNF-α, IL-6 and IL-8 levels decreased between 56-81% in subjects treated with 70 mg or 150 mg POLB 001 twice daily
†The exploratory analysis suggested statistically significant improvement in treatment (p<0.05) for the endpoints examined.
Reduced Key Indicators of LPS-Induced Systemic Inflammation
The reduction of systemic cytokines align with improvement in clinically meaningful endpoints

Mean Body Temperature

No significant effect on body temperature with a trend towards reduction compared to placebo
Heart Rate Rise (bpm)

Suppressed increase in heart rate following IV LPS administration
C-Reactive Protein (CRP)

CRP level reductionof 33.1% and 33.3% seen for 70mg and 150mg doses respectively

POLB 001 Prevented CRS in Humanised Mouse Model
Highly effective and superior to a TNF-α antibody in a gold standard model of CRS


CRS Scores
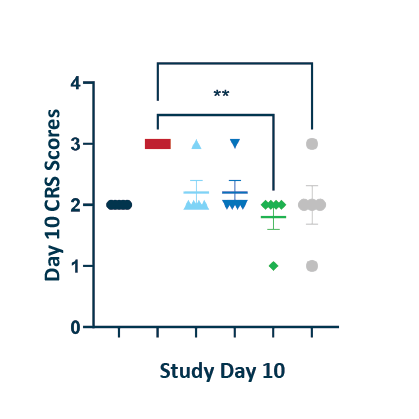
POLB 001 prevented CRS symptoms*
CRS Biomarkers
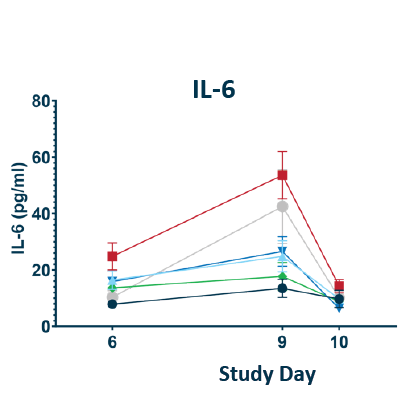
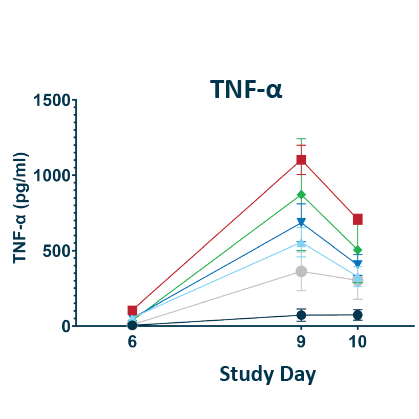
POLB 001 decreased all key CRS biomarkers tested
The experimental model is a previously validated CD28 superagonist induced CRS model in humanized tumour bearing mice performed by The Jackson laboratory. A TNF antibody was included as a robust comparator as these have been found empirically to be the most potent preventors of CRS in mice despite limited utility in humans. *Statistically significant reduction of CRS scores compared to untreated controls. CRS scores had no significant difference to No CRS Control group. BID: twice daily; CRS: Cytokine Release Syndrome; TNF: Tumour necrosis factor; IL-6: Interleukin-6; IL-8: Interleukin-8
Strong Patent Portfolio & Orphan Drug Designation
- FDA Orphan Drug Designation granted in May 2025 for POLB 001 for prevention of bispecific antibody-induced CRS
- March 2026 – Major milestone – first national grant within oncology CRS patent family received from IP Australia
- Oncology patent applications with potential for protection out to at least 2044; in vivo animal data enhances & facilitates expansion of patent applications
- Significant US patent granted in 2024 covering a class of drugs (including POLB 001) for treating hypercytokinaemia (cytokine storm) and for preventing hypercytokinaemia in a patient after an immune response has been triggered. This encompasses cytokine storm that is induced in any disease indication
- Granted patents for severe influenza which provide protection until at least 2038
TOPICAL Clinical Trial
Trial of Prevention of ImmunoCytokine Adverse events in Myeloma
Trial designed to produce rapid & compelling data for the effectiveness of POLB 001 to prevent CRS – Interim data expected summer 2026
| Chief Investigator: Dr Emma Searle, MBChB MA MRCP FRCPath PhD | Trial run by: Accelerating Clinical Trials (ACT) – specialist blood cancer trials organisation |
| Sites: NHS Trusts at The Christie, The Royal Marsden, University College London Hospitals, and University Hospitals Birmingham, as well as NHS Lothian in Edinburgh and the Royal Stoke University Hospital. | Objective: To investigate the safety and efficacy of POLB 001, in particular its ability to reduce incidence of CRS in patients receiving an approved bispecific antibody |
| No. subjects: c. 30 | Patient population: Relapsed/refractory multiple myeloma patients |
“I have seen first-hand the challenges that CRS presents to the delivery of cancer immunotherapies, requiring many of our patients to be hospitalised for treatment. These transformative therapies will continue to be restricted until there is a way to administer them more safely. POLB 001 holds great promise in tackling this issue; potentially leading to improved patient wellbeing, reducing the strain on healthcare systems while making these treatments more accessible to a broader patient population.”
Dr Emma Searle, Consultant Haematologist
Trial Design

Key Objectives/Endpoints
- Incidence of Grade 2+ CRS
- Incidence of CRS all grades
- Confirm safety and pharmacokinetics
- Tocilizumab usage
References
1. Independent research by Decisive Consulting Limited. 2. Average rate from Summary of Product Characteristics (SmPCs) for Yescarta, Tecartus, Abecma, Kymriah, Carvykti, Breyanzi, Elrexfio, Columvi, Epkinly, Tecvayli and Talvey; 3. In this context, adequately is defined as both not completely preventing grade 2+ CRS and potentially sufficient to support active clinical development towards a regulatory approval of a medicine. Grade 2 CRS is defined as described by Lee et al, Biol Blood Marrow Transplant . 2019 Apr;25(4):625-638. janssenscience.com & doi.org/10.1182/blood-2022-159381; CAR T: Chimeric Antigen receptor T cell; 4. Grand View Research. CAR T-Cell Therapy Market Analysis 2023-2030. 5. Grand View Research. Bispecific Antibodies Market Size, Share & Trends Analysis Report. 6. Datamonitor Healthcare. Forecast: Diffuse Large B-Cell Lymphoma and Multiple Myeloma, 2023. 7. Datamonitor Healthcare. Forecast: Diffuse Large B-Cell Lymphoma and Multiple Myeloma, 2023. 8. American Society of Cancer, 2025. 9. Keesari, P.R., Samuels, D., Vegivinti, C.T.R. et al. Navigating the Economic Burden of Multiple Myeloma: Insights into Cost-effectiveness of CAR-T and Bispecific Antibody Therapies. Curr Hematol Malig Rep 20, 3 (2025). 10. Datamonitor Healthcare. Forecast: Diffuse Large B-Cell Lymphoma and Multiple Myeloma, 2023. 11. An average of Badaracco et al. (2022), McGarvey et al. (2023), and Locke et al.




